A new preclinical study from Weill Cornell Medicine found that the protein caspase-5 (CASP5), long thought to be a foot soldier in the body’s defense against bacterial infection, does not actually help clear invaders the way its close cousin caspase-4 (CASP4) does. Instead, the researchers discovered that CASP5 boosts the signal to proliferate in a population of gut cells that maintains a healthy intestinal lining and replaces injured cells with new, healthy ones.
The study, published April 22 in Nature, identifies CASP5 as a previously unrecognized “switch” that helps control tissue regeneration and maintenance. Disruptions in this system are linked to diseases like colorectal cancer and inflammatory bowel disease (IBD), which destroys the lining of the digestive tract and causes diarrhea, stomach pain and nausea.
“We’ve shown that a specific form of caspase-5 is a powerful enzymatic regulator of the Wnt signaling pathway, the master control system that tells cells when to grow and divide,” said Dr. Julie Magarian Blander, the Gladys and Roland Harriman Professor of Immunology in Medicine and a member of the Jill Roberts Institute for Research in Inflammatory Bowel Disease at Weill Cornell.
Co-first authors Drs. Baosen Jia and Yuhua Shi played pivotal roles in the biochemical, organoid and functional experiments that led to this discovery.
Different Forms of CASP5 with Different Functions
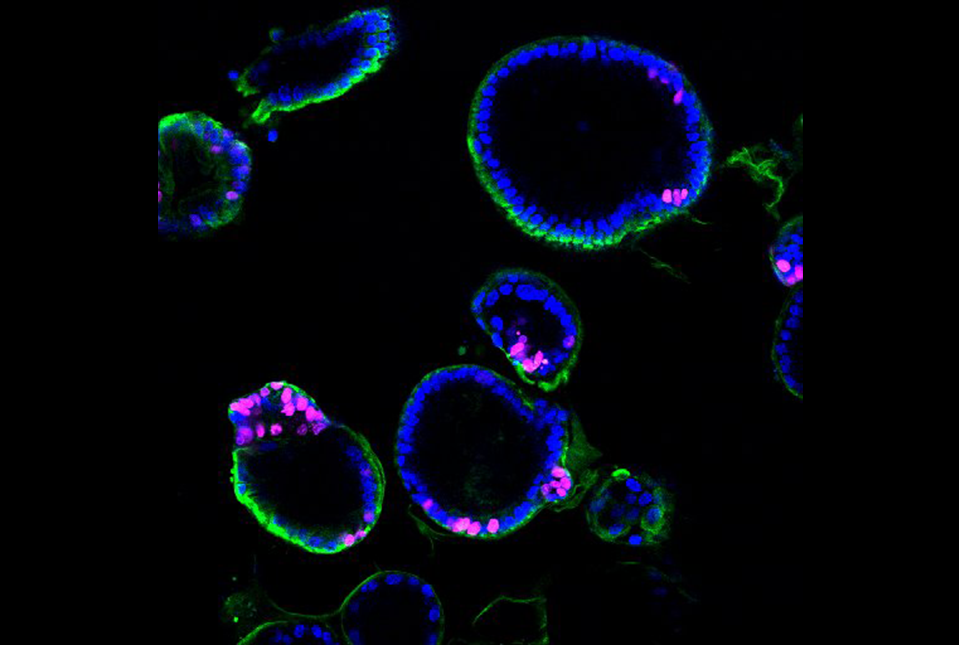
The team created human colonic and small intestinal organoids in the lab to study changes in CASP5 throughout the differentiation of immature stem cells to the fully functioning epithelial cells that make up the gut’s lining. These small balls of cells were made from human intestinal or colonic stem cells.
Through a series of experiments, the team discovered that the CASP5 protein exists in three different forms, or isoforms, inside human intestinal epithelial cells. The shortest form, called CASP5c, is found mainly in a critical zone of the intestinal crypts where new cells are born and begin their journey upward. These are the transit-amplifying cells, the “workhorse” cells that must rapidly multiply before they mature into the cells that line the gut.
CASP5c turns out to be an amplifier of Wnt signaling. It does this by cutting a key protein called APC inside the cell. When APC is cut, the brakes on Wnt signaling are released, allowing another protein called β-catenin to accumulate and turn on genes that promote cell proliferation and repair.
The team found that, in contrast, the two longer forms, CASP5a and CASP5b, act like natural brakes on cell proliferation. They compete with CASP5c for the same binding partner, Dishevelled (a hub protein in the Wnt signaling pathway), but cannot cut APC. By occupying the “driver’s seat,” they prevent excessive Wnt activity in the upper parts of the crypt where cells are supposed to differentiate into mature, specialized cells.
“This creates a beautifully balanced system: high levels of CASP5c in the lower crypt push proliferation when it is needed, while rising levels of CASP5a and CASP5b higher up apply the brakes, so cells mature properly,” Dr. Blander said. “It is as if the gut has its own internal dimmer switch that fine-tunes proliferation and repair exactly where and when it is required.”
Disease Connection
This discovery is especially important for IBD. When Dr. Blander’s team examined intestinal samples from patients with ulcerative colitis or Crohn’s disease, they found elevated levels of CASP5c. “With caspase-5c levels so high, we felt that transit-amplifying cells were working hard to restore the epithelium in these patients,” Dr. Blander said. “The protein essentially nurses these progenitor cells, making sure they have the proliferative boost they need during injury.”
This finding suggests that the body naturally turns up this repair switch during injury. “If the balance between the isoforms is disrupted, for example, if CASP5c stays too active, for too long in a chronically inflamed environment, Wnt signaling could become excessive,” Dr. Blander said. “Potentially, this could lead to abnormal cell growth and long-term risk of dysplasia or colorectal cancer, a known complication of longstanding IBD.”
“This study, together with my lab’s previous work on the inflammasome, shows that many proteins involved in innate immunity have overlapping roles in basic tissue homeostasis,” said Dr. Blander. “Caspase-5 was expected to function in innate host defense against infection, yet here we show it has a critical function in homeostasis of the intestinal epithelium.”
The discovery was the result of a dedicated team effort at Weill Cornell Medicine. Dr. Chongbo Yang and Mr. Dylan Roycroft in Dr. Blander’s team, GI oncologist and Professor, Dr. Sabine Tejpar at KU Leuven in Belgium and bioinformatician Dr. Yourae Hong (working with Dr. Tejpar’s team) helped map the spatial patterns and clinical relevance of CASP5 isoforms. Dr. Steven Lipkin, formerly the Gladys and Roland Harriman Professor of Immunology in Medicine at Weill Cornell Medicine, provided valuable human organoids. The work also benefited greatly from the Weill Cornell Medicine Microscopy and Image Analysis and the Human Therapeutic Organoid Cores, as well as the Center for Translational Pathology and the Live Cell Bank, which supplied essential human tissue samples and organoids.
Many Weill Cornell Medicine physicians and scientists maintain relationships and collaborate with external organizations to foster scientific innovation and provide expert guidance. The institution makes these disclosures public to ensure transparency. For this information, please see the profile for Dr. Julie Magarian Blander.
This work was supported by Weill Cornell Medicine start-up funds; the National Institutes of Health through grant numbers AI170832, AI170897, AI159772 and AI178327; the Binational US-Israel Science Foundation; the Kenneth Rainin Foundation; the National Cancer Institute through grant U54-CA272688; the Crohn’s & Colitis Foundation; and the Burroughs Wellcome Fund.



